What is petroleum anyway?
Petroleum is a natural product consisting of long-chain hydrocarbons, sulfur and sulfur compounds, metals and rocks [22]. It arises when organic material e.g. Plants or animals in the upper crust are exposed to elevated pressures and temperatures. Today's crude oil mainly consists of so-called dead marine creatures, microplankton, algae and small animals, which can only be seen under the microscope [21]. It is extracted from rock layers, refined and passed on to consumers. It has been known to us humans for more than a thousand years, under the name of Petroleum, which is composed of the Greek words petra - rock / rock and oleum - oil, which is why it is also called rock oil.
How is oil produced?
Crude oil is formed when dead marine minerals in the upper crust are compressed and heated. It converts the carbohydrates, proteins and lipids contained in the animals into long-chain hydrocarbons. The more pronounced the factors of pressure and temperature are, the more the composition of the crude oil changes. From a temperature of 60 ° C arise in some cases short-chain hydrocarbons such as gaseous methane (also called marsh gas), ethane (which is used as a heating agent), propane and butane (which are contained in gas burners), but also liquid pentane, hexane, heptane and octane which are used as fuels for cars, for heating and for energy production. Between 120-130 ° C arise for the most part, and from 200 ° C arise only short-chain hydrocarbons.
How do we get oil?
The oil is in the upper crust of the earth, which is why we need to drill afterwards. This happens in three stages:
1. The hole
The primary extraction, which uses the natural pressure of oil deposits to bring oil to the surface.
2. The pumping
The secondary production, where water or gas is pumped into the borehole to get petroleum residues to the surface
3. Collect the remains
The tertiary promotion, this steam, polymers, CO2 or chemicals are used to get hold of last remains in the occurrence.
Also drilling rigs are used for sea drilling, these act on the same principles.
What are the problems with oil and its gathering?
Oil production releases vast amounts of greenhouse gases while pumping chemicals and non-biodegradable plastics into the soil and thus into our groundwater. Greenhouse gases such as methane are released by the extraction and refining of crude oil. Sulfur-containing crude, e.g. releases large amounts of SO2 (sulfur dioxide), which in the atmosphere is converted to sulfurous acid and sulfuric acid, which manifests itself as "acid rain". In addition, there are often petrol disasters that irreversibly affect the environment. Leaking oil from drilling rigs, or sinking cargo ships carrying tons of oil on board, released about 100,000 [1] thousand tons of oil into our seas each year. This has catastrophic consequences for the entire planet. The best example of this is the "oil spill" in the Gulf of Mexico, which resulted in follow what still noticeable on the Mississippi delta.
Why is petroleum used in cosmetics?
Mineral oil derivatives are used as a base in oils, petrolatum and waxes. They are inexpensive, can be processed in consistent quality, and do not trigger allergies. However, they contain so-called Mineral Oil Aromatic Hydrocarbons, MOAH's short[2].
What consequences do oil-based cosmetics have?
One of the big problems with oil drilling is the fact that radioactive particles, metals and heteroatom compounds, as well as MOAHs, are carried with oil. Long-chain hydrocarbons have little effect on the human body, both positive and negative, but these contaminants already. Metals are among iron, copper, nickel and vanadium. Nickel and vanadium are KMR substances [3], which means they are:
- carcinogenic,
- mutagenic,
-or reprotoxic
Heteroatoms are those organic compounds which have one or more non-carbon (C-C) or hydrogen (C-H) compounds. Such are with under:
Oxygen: Alcohols (such as ethanol for drinking, but also methanol which causes blindness [4]), carboxylic acids (for example vinegar), ketones (for example acetone which is contained in solvents for paints and varnishes, as well as in nail polish removers)
- Nitrogen: Amines and amides (which often smell bad, irritate or are toxic [5]), amino acids (eg, from which our DNA and neurotransmitters, such as dopamine or serotonin, exist [6]), nitro compounds (which eg used in explosives or paint strippers [7])
- sulfur: thiols (which smell horribly [8]), sulphates and sulphides (for example nickel (II) sulphate [9])
- Phosphorus: phosphanes and phosphoric acid esters (highly toxic substances [10])[10])
- Halogens: fluoro-, chloro-, bromo-, and iodoalkanes (eg carbon tetrafluoride, which is used as a refrigerant. [11] Chloroform is a poisonous, volatile liquid. [12] Bromobenzene, which is highly irritating to the respiratory tract. [13] Iodoalkanes, which release free iodine radicals (I-), which in turn attack the skin [14])
However, such particles are mostly removed, only MOAHs are still between eight (in body oils) and 15,000 (in petrolatum) times as high as in food, according to independent tests.[15]
Radium compounds such as sulfates and carbonates are also carried along with the rock. The main part of these compounds consists of the isotope radium-226 with a half-life of 1,600 years [16]. However, it is unlikely that relevant amounts are present in the crude oil. By contrast, larger amounts of slurries of radium-226 compounds are produced by the separation methods, which are often not or not properly disposed of [17], since there are no clear rules.
Another part of the petroleum, in addition to long-chain hydrocarbons, are cyclic hydrocarbons, and so-called aromatics. They differ in the fact that they form both carbon rings and hexagons, which have aromatics as distinct chemical double bonds.[18]
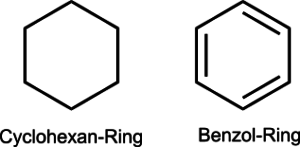
Cyclischer Kohlenwasserstoff Aromat Abb. 2
Hierbei ist anzumerken, dass solche Ringe meist sehr giftig sind. Cyclohexan kann dabei z.B. zu Dermatitis führen[19], während Benzen (Benzol ist ein veralteter Begriff) als Krebserregend gilt[20].
However, once a part, even a small one, changes part of a molecule, it can have a huge impact on its properties. Thus, benzene is e.g. Carcinogenic, cinnamon aldehyde, which "contains" the benzene as the major part, gives cinnamon its characteristic odor and taste, and it is quite harmless.
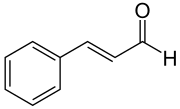
Zimtaldehyd Molekül Abb. 3
Why do not we use any petroleum-based raw materials in our products?
The facts about oil, the pollution of the environment with exhaust gases and waste, as well as the partly strongly varying ingredients are reason enough to look for alternatives. And we find that sufficient in nature. Herbal extracts and ingredients are as environmentally friendly as possible, and often work just as well, if not better and gentler than petroleum derivatives.
To love the environment, and for one's own conscience, such alternatives are the best means.en sind solche Alternativen das beste Mittel.
Source reference for reading and informing:
Radioactive oil slurries [17]
Independent consumer protection tests [2][15]
Methanol poisoning [4]
Benzen, its consequences and uptake ways [20]
KMR list of the Federal Ministry for the Environment, Nature Conservation, Building and Nuclear Safety [3]
Petroleum formation and exploitation [21] [22]
Wikipedia [1][5][6][7][8][9][10][11][12][13][14][16][18][19]
Sources: Wikipedia, Stiftung Warentest, Planet-wissen.de, Noxen.de, Federal Ministry for the Environment, Nature Conservation, Building and Nuclear Safety